Laboratory water is not a single-grade resource; it is a precisely controlled reagent that directly impacts analytical accuracy, experimental reproducibility, and equipment longevity. From routine glassware cleaning to ultra-sensitive trace analysis, different applications require specific levels of purity. As a result, laboratory water purification systems are designed in multiple configurations, each tailored to achieve defined quality standards.

Primary Classification Based on Water Purity Standards
Laboratory water systems are commonly categorized according to internationally recognized standards such as ASTM, ISO, and CLSI. These standards define three main types of purified water: Type I, Type II, and Type III.
Type I Water (Ultrapure Water)
This represents the highest purity level available, typically with resistivity of 18.2 MΩ·cm. Type I water is free from ions, organic compounds, bacteria, and particulates. It is essential for highly sensitive analytical techniques such as HPLC, ICP-MS, and molecular biology experiments. These systems often incorporate advanced technologies such as ion exchange, UV oxidation, and ultrafiltration.
Type II Water (Pure Water)
Type II water is suitable for general laboratory applications including buffer preparation, media preparation, and reagent dilution. While not as pure as Type I, it still maintains low levels of contaminants and is often used as feed water for Type I systems.
Type III Water (RO Water)
Type III water is typically produced using reverse osmosis (RO) and is used for less critical applications such as glassware rinsing or as feed water for higher purification systems. It removes the majority of dissolved solids and organic contaminants but does not reach analytical-grade purity.
Classification Based on Purification Technology
Different purification technologies are combined in laboratory systems to achieve the desired water quality. The selection depends on feed water conditions and required output purity.
Reverse Osmosis (RO) Systems
RO water purification systems use semi-permeable membranes to remove up to 99% of dissolved salts, bacteria, and organic materials. These are commonly used as a pre-treatment stage in laboratory water systems.
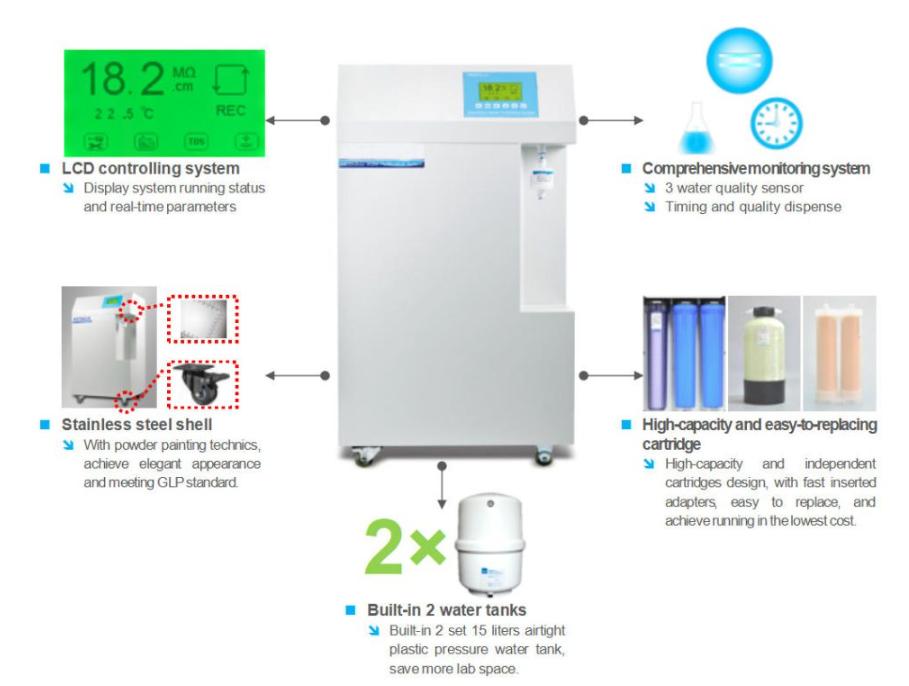
Deionization (DI) Systems
DI systems utilize ion-exchange resins to remove charged particles such as cations and anions. They are effective for producing low-conductivity water but may not remove organic contaminants or microorganisms.
Ultraviolet (UV) Purification Systems
UV systems are used to reduce microbial contamination and break down organic compounds through photo-oxidation. They are often integrated into high-purity (Type I) systems.
Ultrafiltration (UF) Systems
UF membranes remove endotoxins, nucleases, and particulates, making them essential in life science applications such as cell culture and molecular biology.
Classification Based on System Configuration
Laboratory water purification systems can also be categorized by how they are installed and distributed within the lab.
Centralized Systems
These systems supply purified water to multiple workstations through a distribution loop. They are ideal for large laboratories or facilities with high water demand, ensuring consistent quality across all outlets.
Point-of-Use Systems
Installed directly at the workstation, these systems provide immediate access to purified water. They are commonly used for producing Type I water close to sensitive instruments.
Modular Systems
Modular systems allow laboratories to customize purification stages according to their needs. This flexibility is beneficial when upgrading existing setups or adapting to new applications.
Integrated and Smart Water Purification Systems
Modern laboratory water purification systems increasingly incorporate automation and digital technologies. Smart systems feature real-time monitoring of parameters such as resistivity, TOC (Total Organic Carbon) and flow rate. Integration with IoT platforms allows remote diagnostics, predictive maintenance, and automated alerts, significantly improving operational efficiency and reducing downtime.
These systems are particularly valuable in regulated environments, where data traceability and compliance with quality standards are critical.

Summary
Laboratory water purification systems are diverse and highly specialized, reflecting the wide range of scientific applications they support. Whether classified by purity level, purification technology, or system configuration, each type serves a distinct role in ensuring reliable laboratory performance. Selecting the right system requires careful consideration of application needs, feed water quality, and operational demands, ultimately ensuring that water quality never becomes a limiting factor in scientific progress.